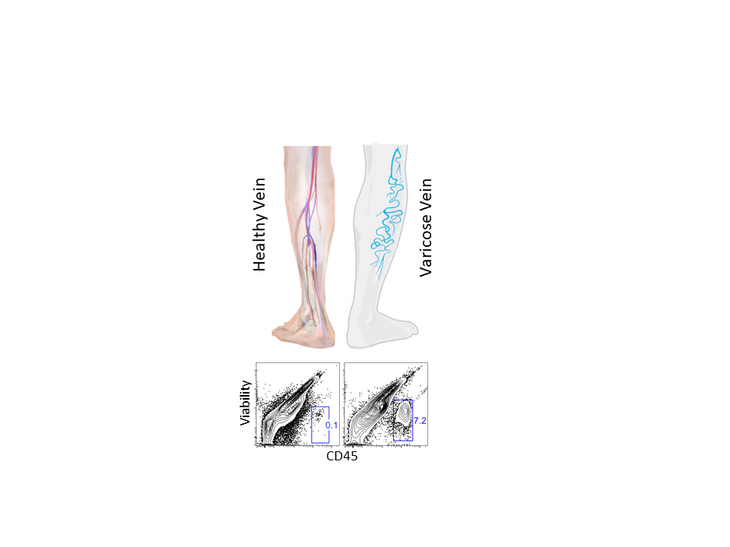
Inflammatory Monocyte Counts Determine Venous Blood Clot Formation and Resolution.
Shahneh F, Christian Probst H, Wiesmann SC, A-Gonzalez N, Ruf W, Steinbrink K, Raker VK, Becker C.
Arterioscler Thromb Vasc Biol. 2021 Dec 16:ATVBAHA121317176. doi: 10.1161/ATVBAHA.121.317176.
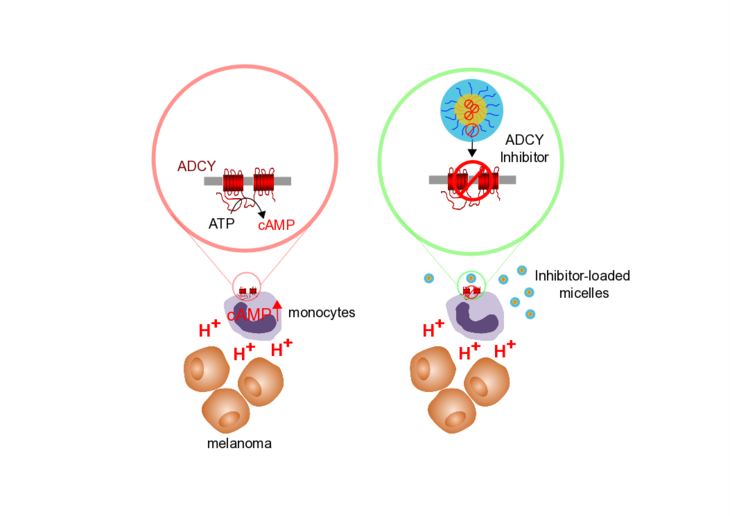
Therapeutic melanoma inhibition by local micelle-mediated cyclic nucleotide repression.
Johann K, Bohn T, Shahneh F, Luther N, Birke A, Jaurich H, Helm M, Klein M, Raker VK, Bopp T, Barz M, Becker C.
Nat Commun. 2021 Oct 13;12(1):5981. doi: 10.1038/s41467-021-26269-w.
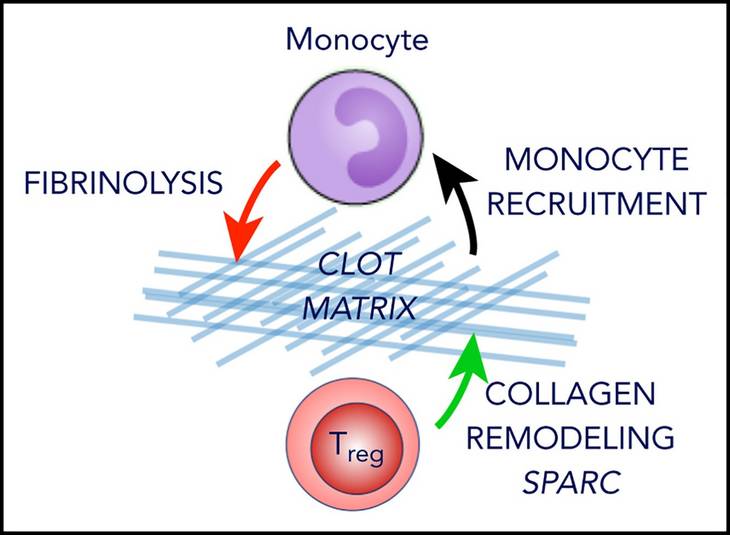
Specialized regulatory T cells control venous blood clot resolution through SPARC.
Shahneh F, Grill A, Klein M, Frauhammer F, Bopp T, Schäfer K, Raker V, Becker C.
Blood. 2020 Sep 15:blood.2020005407. doi: 10.1182/blood.202000540
Thrombo-Inflammation in Cardiovascular Disease: An Expert Consensus Document from the Third Maastricht Consensus Conference on Thrombosis.
d'Alessandro E, Becker C, Bergmeier W, Bode C, Bourne JH, Brown H, Buller HR, Ten Cate-Hoek AJ, Ten Cate V, van Cauteren YJM, Cheung YFH, Cleuren A, Coenen D, Crijns HJGM, de Simone I, Dolleman SC, Klein CE, Fernandez DI, Granneman L, van T Hof A, Henke P, Henskens YMC, Huang J, Jennings LK, Jooss N, Karel M, van den Kerkhof D, Klok FA, Kremers B, Lämmle B, Leader A, Lundstrom A, Mackman N, Mannucci PM, Maqsood Z, van der Meijden PEJ, van Moorsel M, Moran LA, Morser J, van Mourik M, Navarro S, Neagoe RAI, Olie RH, van Paridon P, Posma J, Provenzale I, Reitsma PH, Scaf B, Schurgers L, Seelig J, Siegbahn A, Siegerink B, Soehnlein O, Soriano EM, Sowa MA, Spronk HMH, Storey RF, Tantiwong C, Veninga A, Wang X, Watson SP, Weitz J, Zeerleder SS, Ten Cate H; Scientific Reviewer Committee.
Thromb Haemost. 2020 Apr;120(4):538-564. doi: 10.1055/s-0040-1708035.
Acute deep vein thrombosis suppresses peripheral T cell effector function.
Prochaska JH, Luther N, Brähler M, Schulz A, Hermanns MI, Lackner KJ, Espinola-Klein C, Münzel T, Wild PS, Becker C.
Br J Haematol. 2019 Mar;184(5):847-850. doi: 10.1111/bjh.15192.
Tumor immunoevasion via acidosis-dependent induction of regulatory tumor-associated macrophages.
Bohn T, Rapp S, Luther N, Klein M, Bruehl TJ, Kojima N, Aranda Lopez P, Hahlbrock J, Muth S, Endo S, Pektor S, Brand A, Renner K, Popp V, Gerlach K, Vogel D, Lueckel C, Arnold-Schild D, Pouyssegur J, Kreutz M, Huber M, Koenig J, Weigmann B, Probst HC, von Stebut E, Becker C, Schild H, Schmitt E, Bopp T.
Nat Immunol. 2018 Dec;19(12):1319-1329. doi: 10.1038/s41590-018-0226-8.
Innate Effector-Memory T-Cell Activation Regulates Post-Thrombotic Vein Wall Inflammation and Thrombus Resolution.
Luther N, Shahneh F, Brähler M, Krebs F, Jäckel S, Subramaniam S, Stanger C, Schönfelder T, Kleis-Fischer B, Reinhardt C, Probst HC, Wenzel P, Schäfer K, Becker C.
Circ Res. 2016 Dec 9;119(12):1286-1295. doi: 10.1161/CIRCRESAHA.116.309301.